 2 nd orbit (energy level) is represented as L shell and it can hold up to 8 electrons.1 st orbit (energy level) is represented as K shell and it can hold up to 2 electrons.However, when an electron loses energy it moves from higher to lower energy level. In an atom, the electrons move from lower to higher energy level by acquiring the required energy. The change in energy occurs when the electrons jump from one energy level to other.Learn the concept of Valency here in detail here. The lowest energy level of the electron is called the ground state. The different energy levels or orbits are represented in two ways such as 1, 2, 3, 4… or K, L, M, N….Learn the concept of an Atomic number here. The range of quantum numbers may vary and begin from the lowest energy level (nucleus side n=1) to the highest energy level. The different energy levels are denoted by integers such as n=1 or n=2 or n=3 and so on.The electrons will not radiate energy as long as they continue to revolve around the nucleus in the fixed orbital shells. Every circular orbit will have a certain amount of fixed energy and these circular orbits were termed orbital shells.The orbits are termed as “stationary orbit.”.Electrons revolve around the nucleus in a fixed circular path termed “orbits” or “shells” or “energy level.”.He concluded that electron will have more energy if it is located away from the nucleus whereas electrons will have less energy if it located near the nucleus.īohr’s Model of an Atom (Source Credit: Britannica) Postulates of Bohr Atomic Model What is Bohr’s Model of an Atom?Īccording to the Bohr Atomic model, a small positively charged nucleus is surrounded by revolving negatively charged electrons in fixed orbits. He explained about electrons and the different energy levels associated with them. Therefore, Rutherford basically explained a nucleus of an atom whereas Bohr took the model one step ahead. 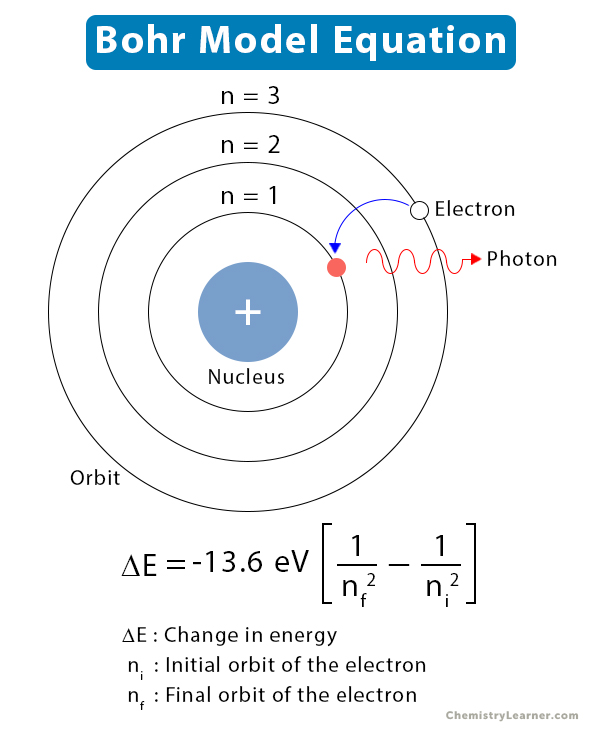
Furthermore, he explained that each orbital shell has fixed energy levels. 
Bohr modified Rutherford’s model where he explained that electrons move around in fixed orbital shells. The electrons move around in a predictable path called orbits. Learn about Rutherford’s Atomic Model here in detail. Earlier in Rutherford Model, Rutherford explained in an atom a nucleus is positively charged and is surrounded by electrons (negatively charged particles). He modified the problems and limitations associated with Rutherford’s model of an atom. Introduction to Bohr Atomic ModelĪ Danish physicist named Neil Bohr in 1913 proposed the Bohr atomic model. German-born physicists James Franck and Gustav Hertz obtained experimental evidence of the presence of these states. The model explained how an atom absorb or emit radiation when electrons on subatomic level jump between the allowed and stationary states. The physicist Niels Bohr said, “Anyone who is not shocked by quantum theory has not understood it.” He also said, “We must be clear that when it comes to atoms, language can only be used as in poetry.” So what exactly is this Bohr atomic model? Let us find out!īohr atomic model and the models after that explain the properties of atomic electrons on the basis of certain allowed possible values. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
